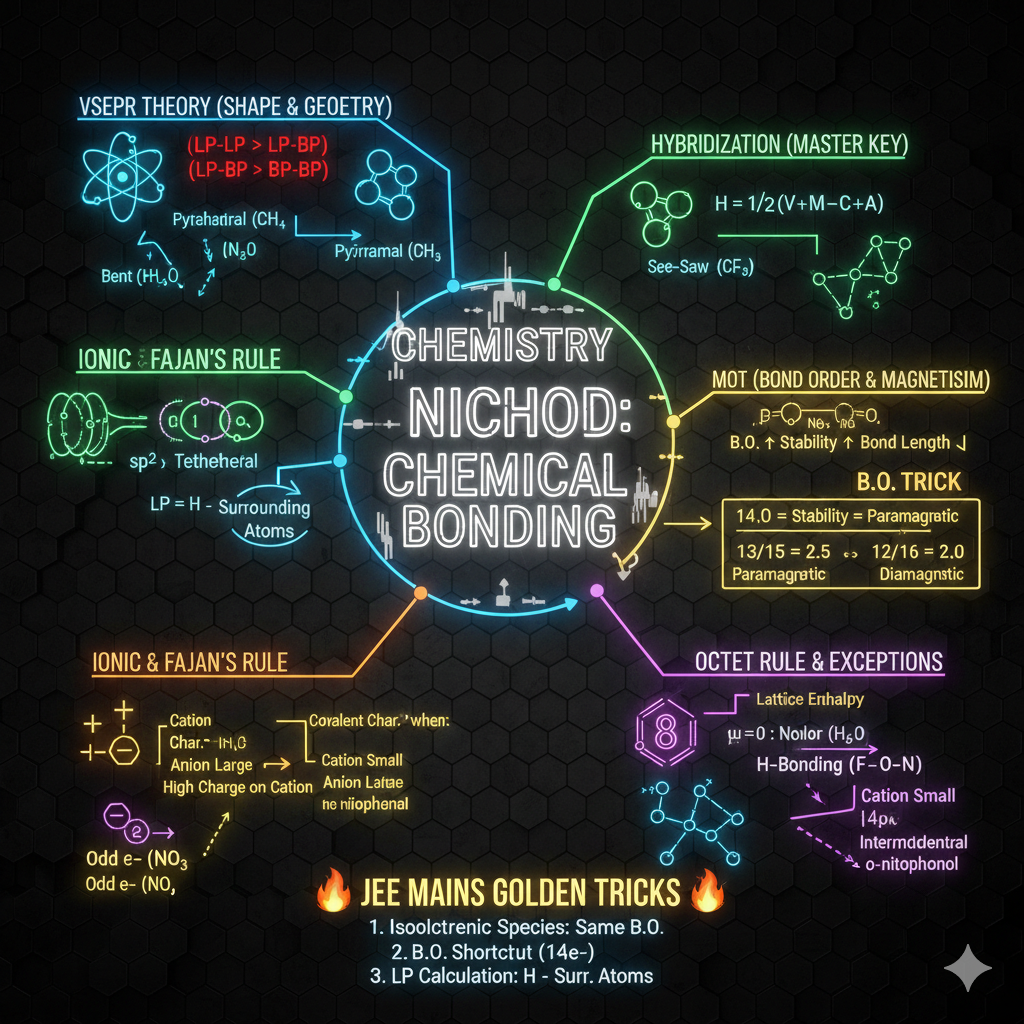
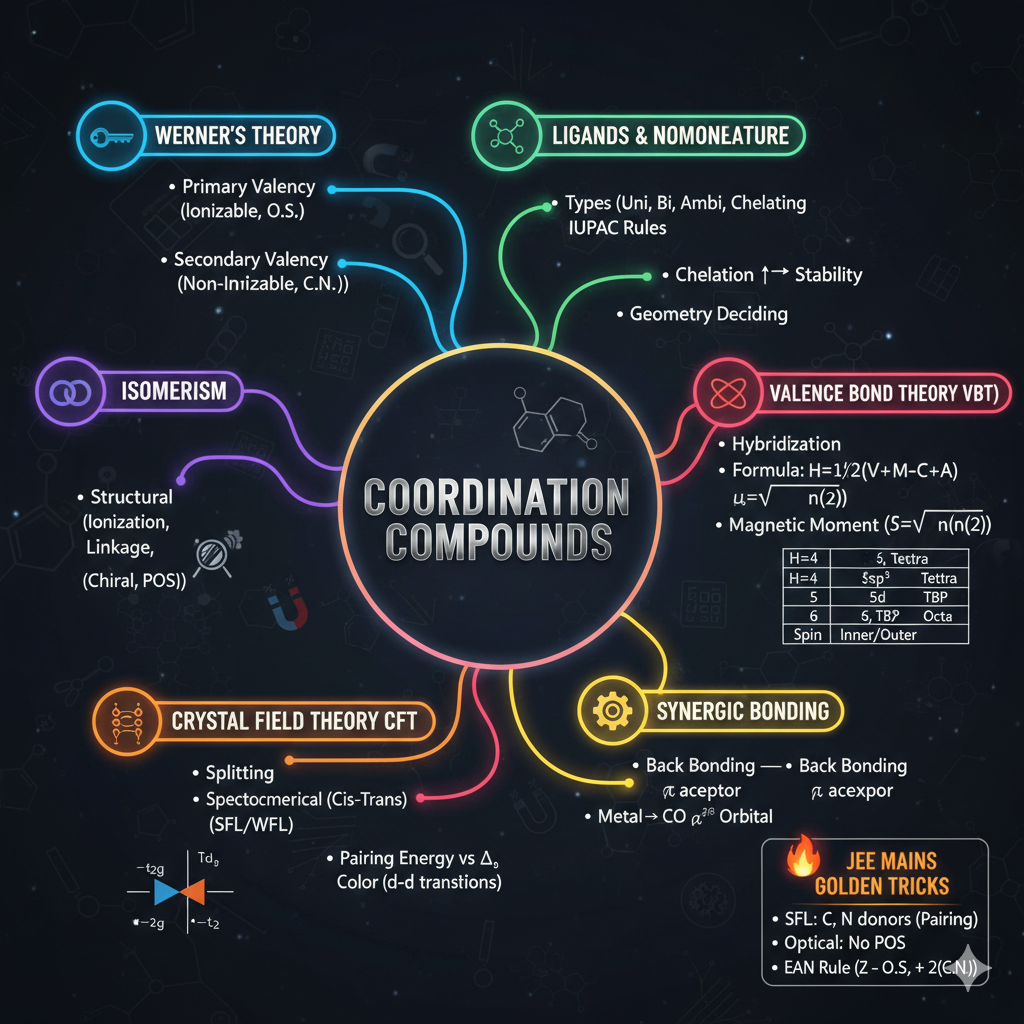
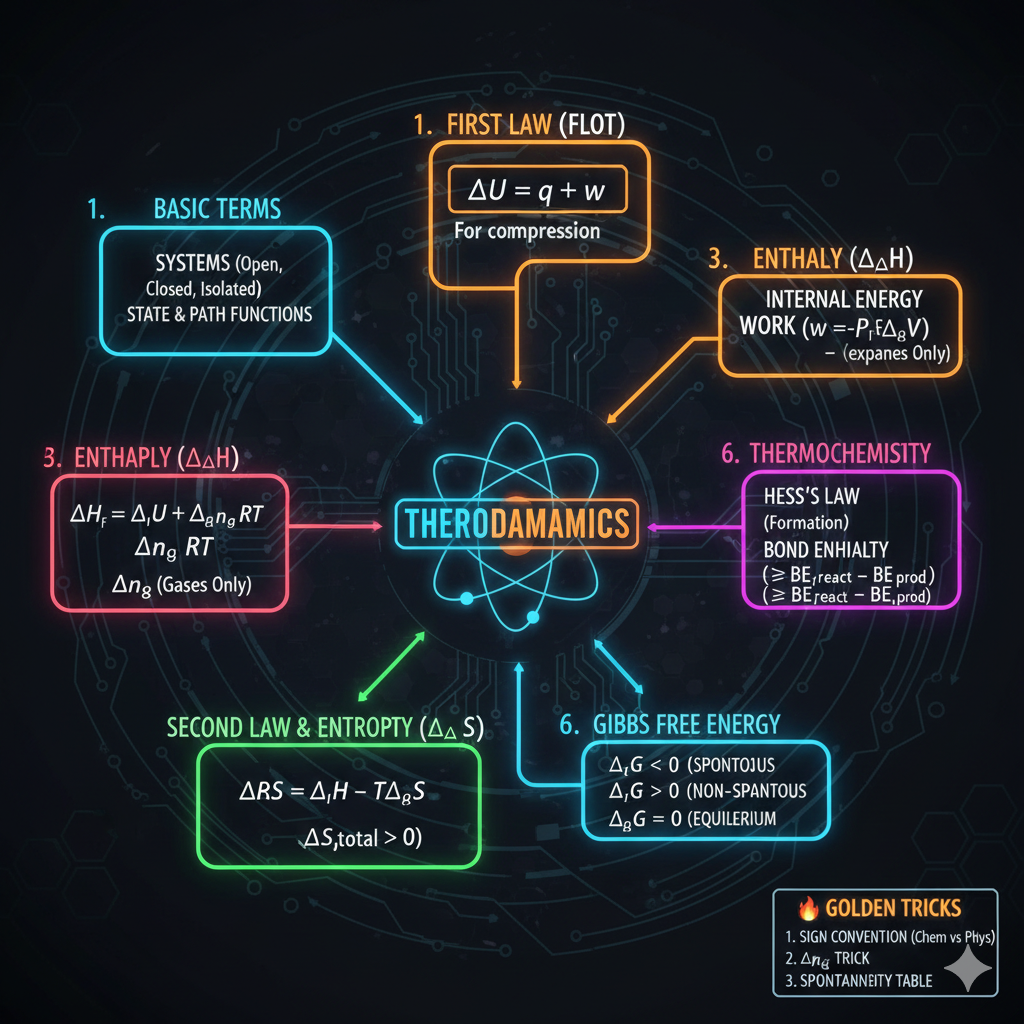
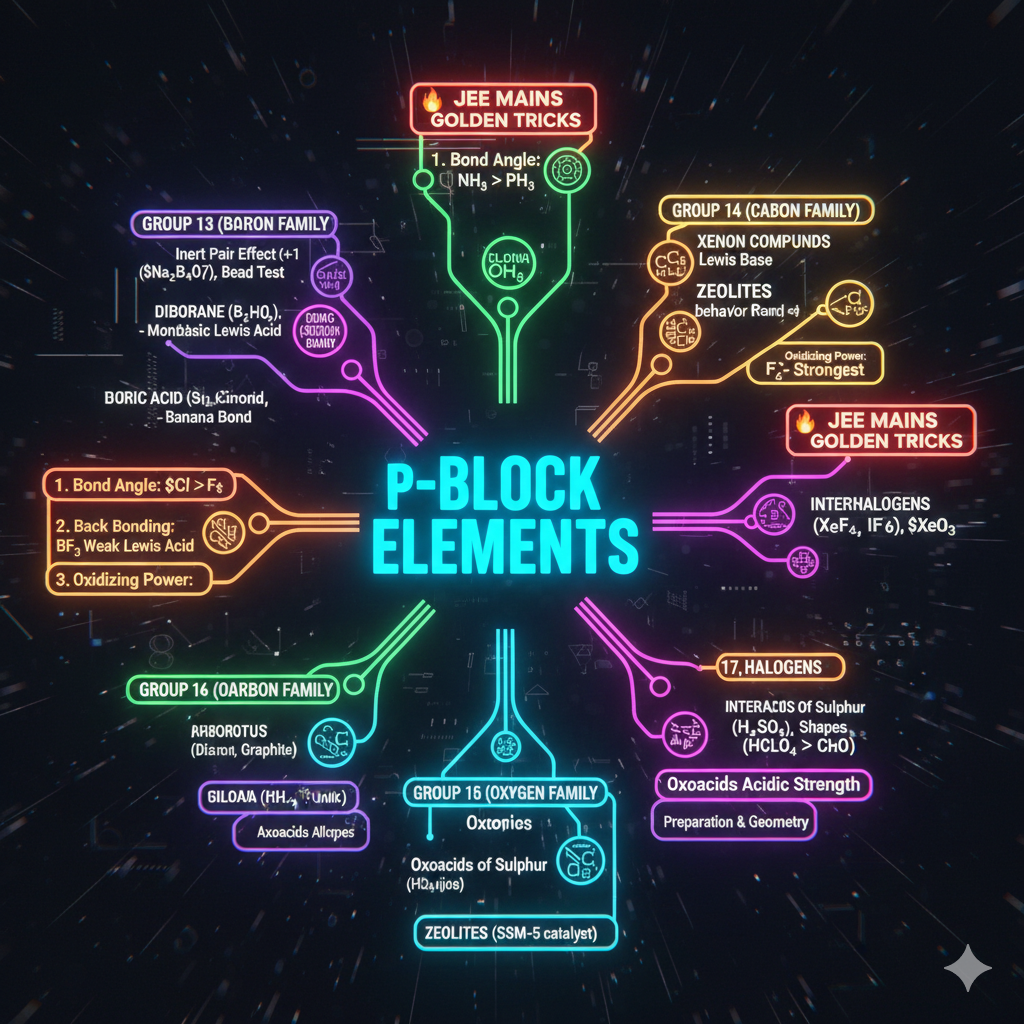
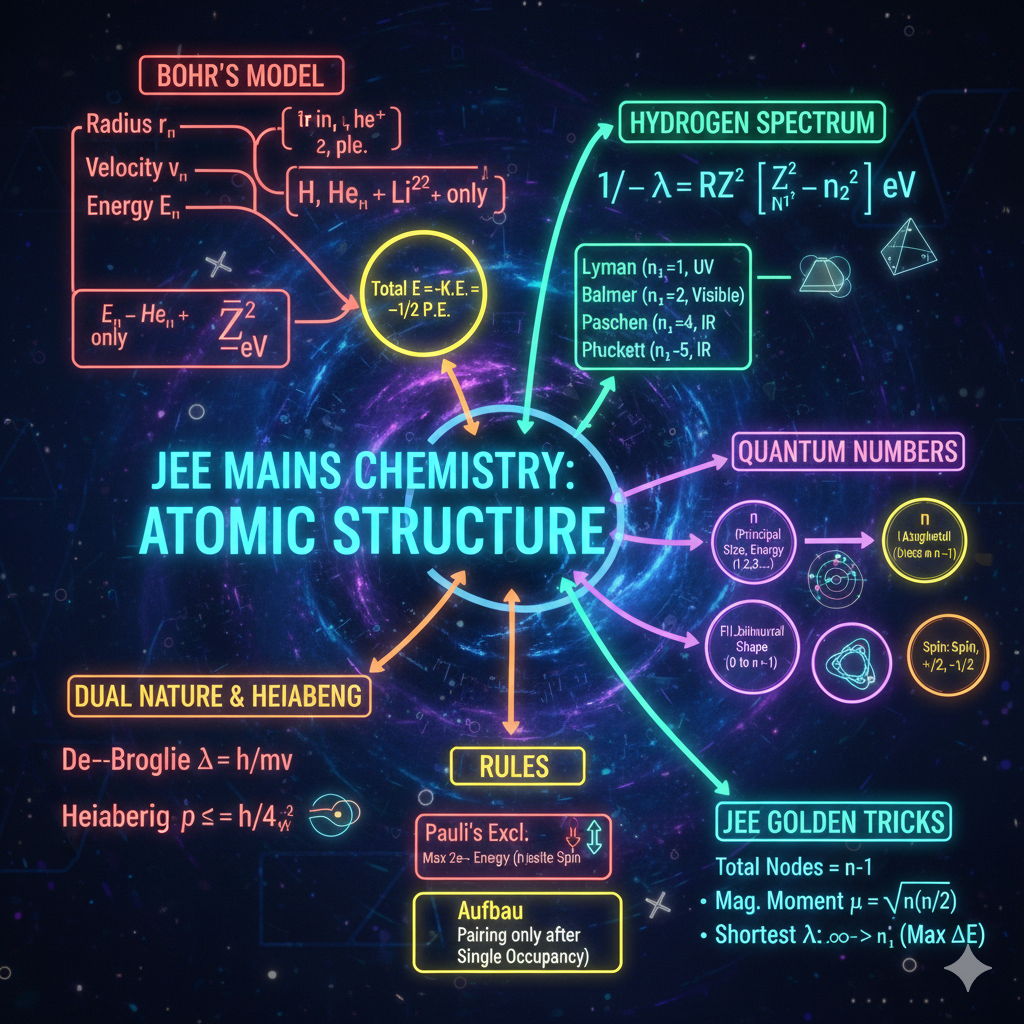
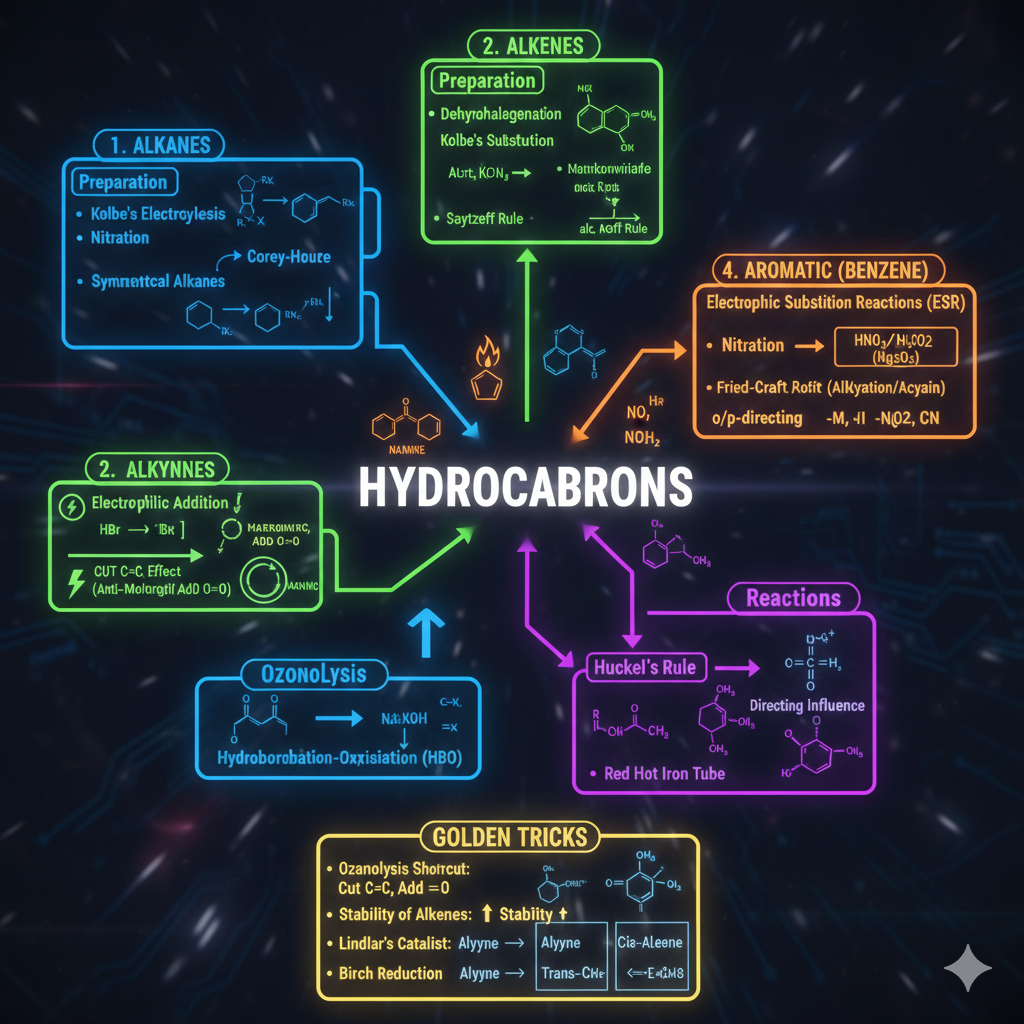
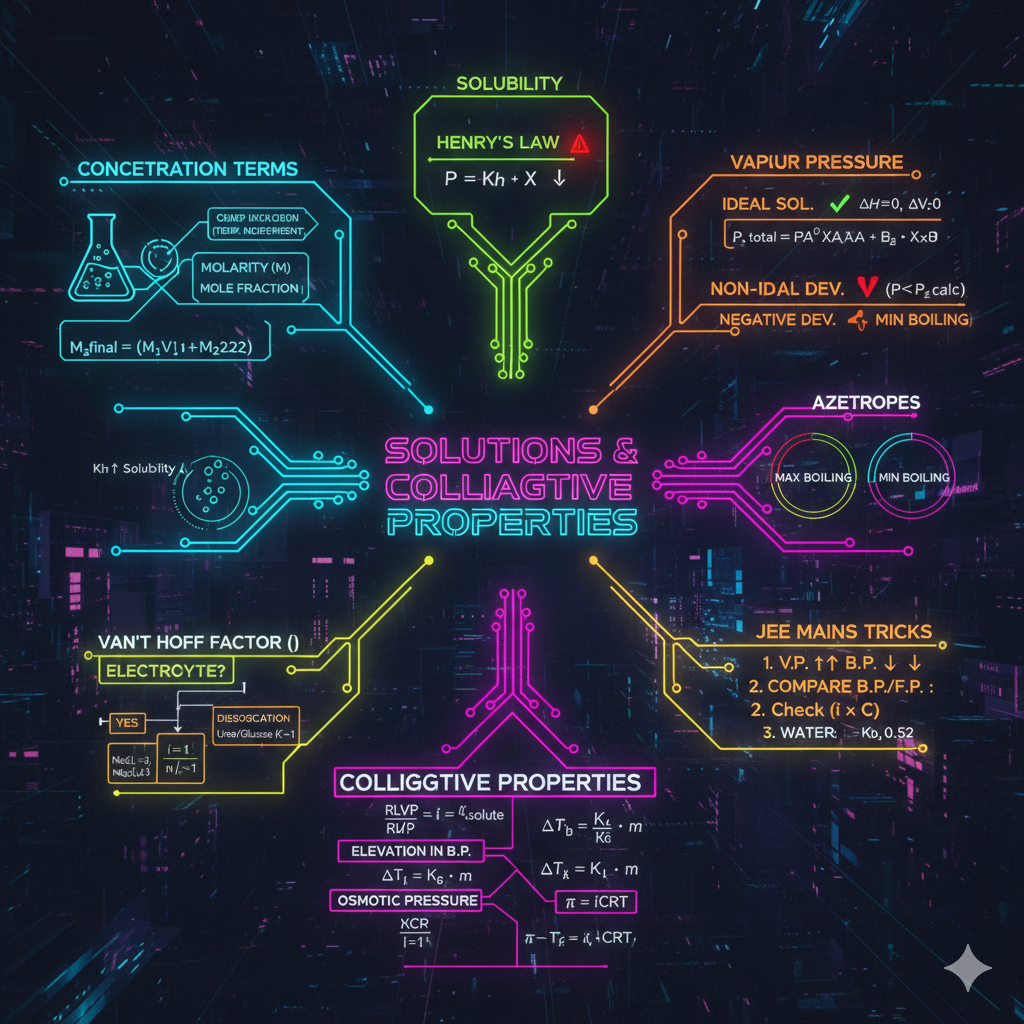
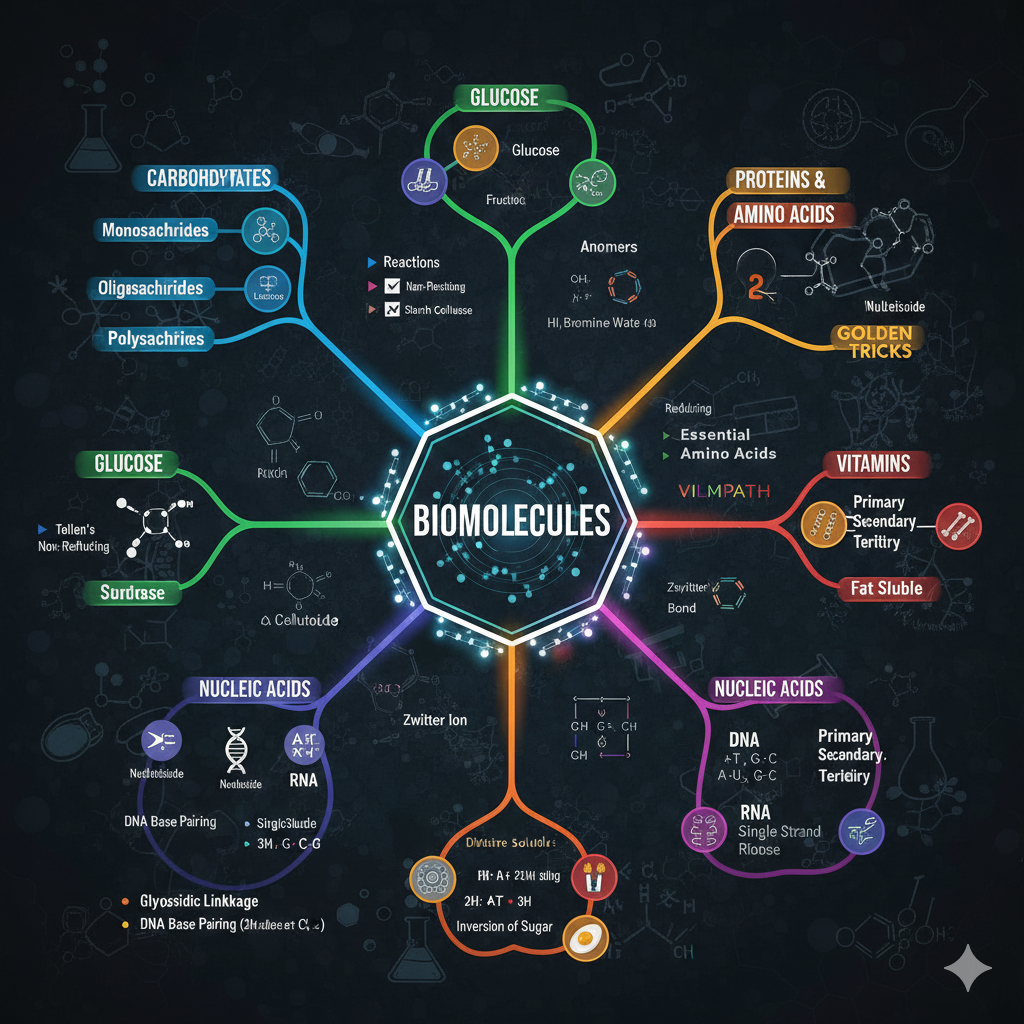
1. ELECTRONIC EFFECTS (Sabse Important)
Ye effects decide karte hain ki molecule kitna reactive ya stable hoga.
- 🟢 Inductive Effect (I): $\sigma$-electrons ka displacement.
(+I: Alkyl groups | -I: $NO_2, CN, F, COOH$) - 🟢 Resonance (M): $\pi$-electrons ka delocalization.
(+M: $OH, NH_2, OR$ | -M: $C=O, NO_2, CN$) - 🟢 Hyperconjugation: $\sigma$-bond (C-H) aur $\pi$-orbital ka overlap. Ise "Baker-Nathan effect" bhi kehte hain.
2. STABILITY OF REACTION INTERMEDIATES
🔹 Carbocation ($C^+$): Resonance > Hyperconjugation > Inductive.
🔹 Carbanion ($C^-$): Stable by -I and -M groups.
🔹 Free Radicals: Follows Carbocation stability pattern.
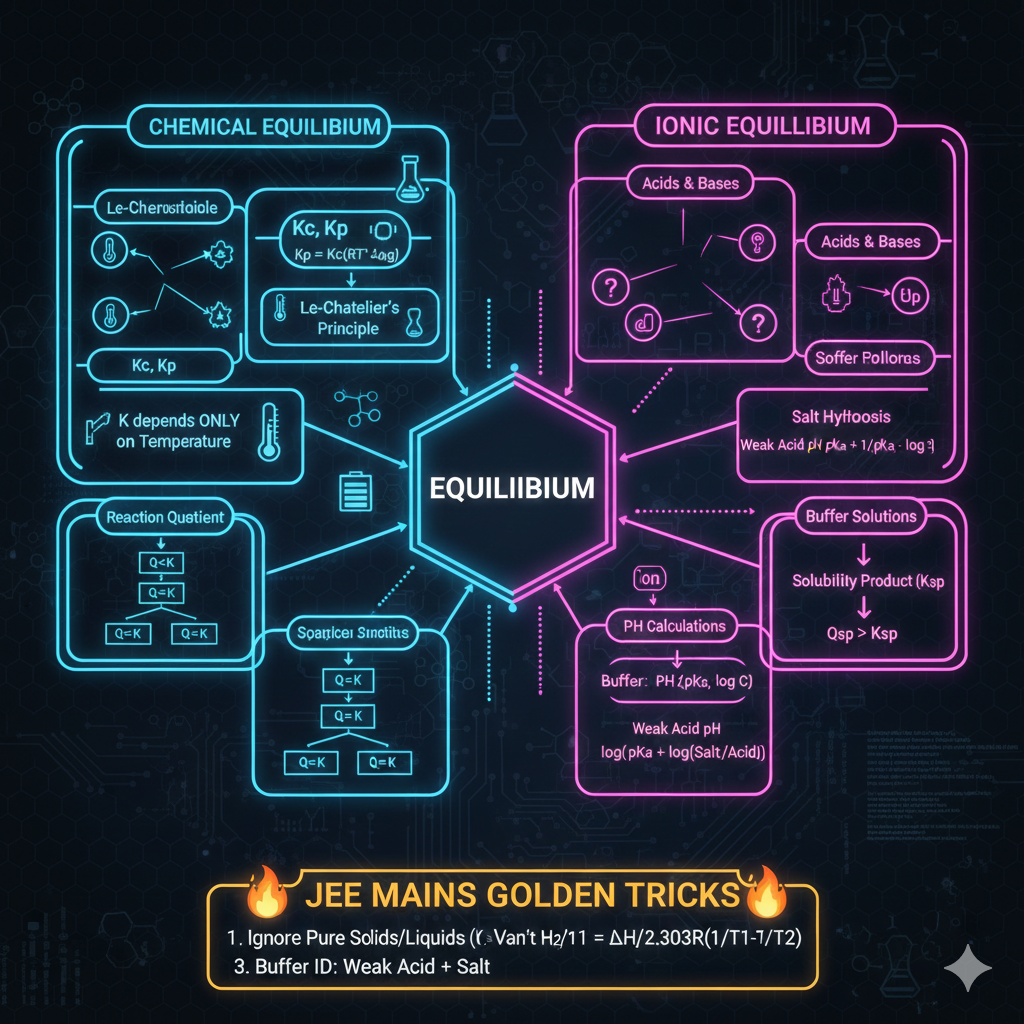
3. ACIDIC & BASIC STRENGTH
Acidic Strength: Directly proportional to -M and -I groups. (Stable anion = Strong Acid).
Basic Strength: Directly proportional to +M and +I groups. (Lone pair availability).
[Image showing acidic strength comparison of substituted phenols]4. ISOMERISM (Short Glance)
• Structural: Chain, Position, Functional, Metamerism, Tautomerism.
• Stereoisomerism: Geometrical (Cis-Trans, E-Z) and Optical (Chiral center, Enantiomers, Meso).
5. REAGENTS & PURIFICATION
Electrophiles ($E^+$): Electron loving ($AlCl_3, BF_3, NO_2^+$).
Nucleophiles ($Nu^-$): Nucleus loving ($OH^-, CN^-, NH_3$).
Analysis: Lassaigne's Test (N, S, Halogens detection) | Kjeldahl’s Method (N estimation).
🔥 JEE MAINS GOLDEN TRICKS
1. Aromaticity: Cyclic, Planar, Fully Conjugated + $(4n+2)\pi$ electrons (Huckel's Rule).
2. Stability Order (A-H-I): Hamesha pehle Aromaticity check karein, fir Resonance (M), fir Hyperconjugation, fir Inductive Effect.
3. Tautomerism: $\alpha$-hydrogen ka hona mandatory hai.
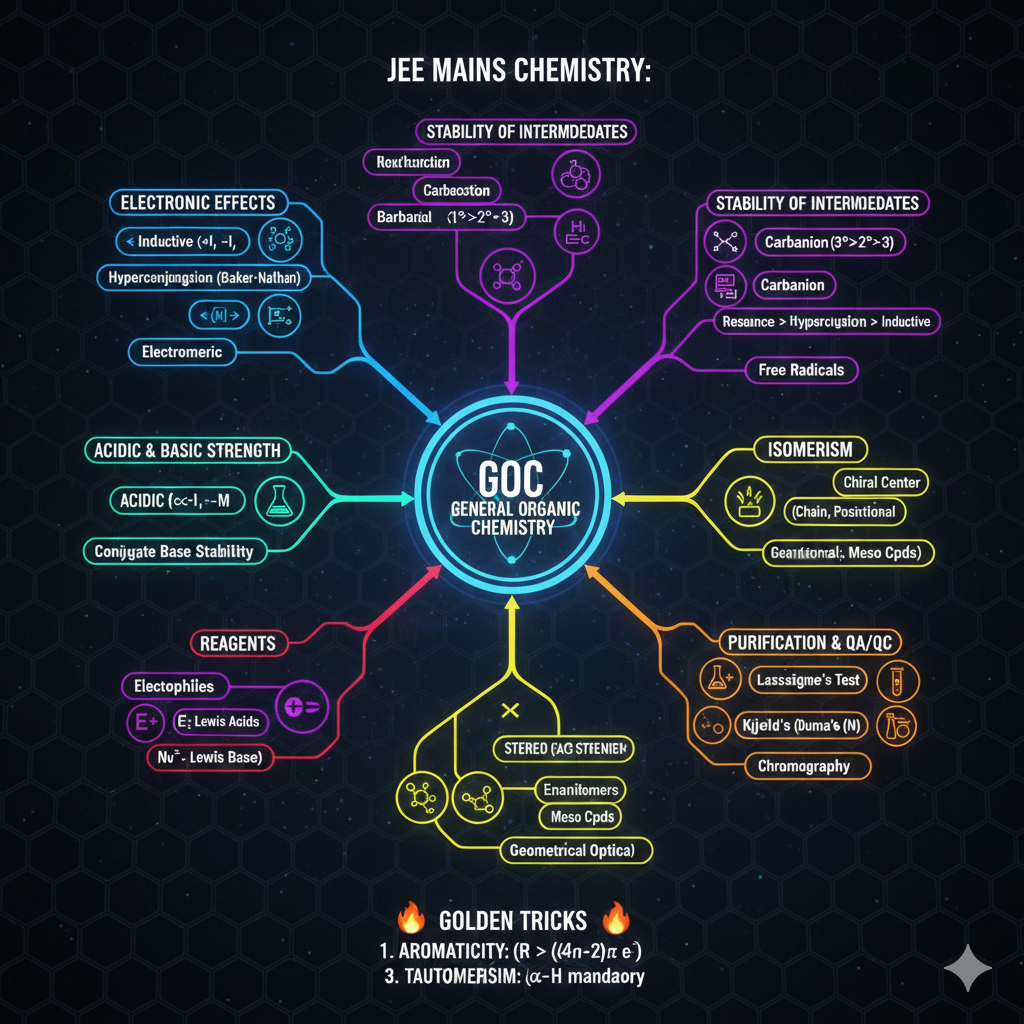
[Image: Electronic Effects and Stability Comparison Table]